Learning objectives
- Name and order the pathways for carbohydrate metabolism of glucose to carbon dioxide during cellular respiration: glycolysis, pyruvate oxidation, and the citric acid cycle
- Identify the major inputs and outputs of each pathway, in terms of carbon compounds, electron carriers, and energy captured by substrate-level phosphorylation of ADP to ATP
- Identify which pathways of carbohydrate metabolism are used for catabolism of proteins and fats
- Differentiate between the locations of the oxidative pathways of glycolysis, pyruvate oxidation, and the citric acid cycle between prokaryotes and eukaryotes
Context and Big Picture
For lots of students, this is the scary part. We’re not going to ask you to try to memorize the steps, the enzymes, or the structures. You can look it up! Instead, you should know the broad outlines, enough to know what to look up. You should especially understand the purpose of each of these pathways and how they connect.
Keep in mind that the main purpose of these pathways is to extract electrons from food molecules to make NADH (so NADH can deliver electrons to the electron transport chain). Secondarily, some of these pathways can also make ATP directly by substrate-level phosphorylation, which becomes important when cells cannot or do not respire.
To understand the connections, follow the carbon atoms.
Before we get into the details, here is the BIG Picture (you’re seen this video before, in the last reading, so you can opt to watch it here for review if you want):
Glucose catabolism begins with glycolysis
All cells must break down glucose into smaller molecules to extract energy for ATP synthesis. Glycolysis means “breaking glucose.” As shown in textbooks, it is a 10-step pathway with 10 enzymes, resulting in two molecules of pyruvate (also called pyruvic acid). All the enzymes of glycolysis are located in the cytoplasm of both prokaryotic and eukaryotic cells.
- Net input: 1 molecule of glucose (6 carbon atoms), 2 NAD+, 2 ADP
- Net output: 2 molecules of pyruvate (3 carbon atoms each), 2 NADH, 2 ATP
- Noteworthy: 2 ATP are hydrolyzed in the initial stages, but 4 ATP are synthesized in the final stages, so there is a net production of 2 ATP
- All ATP synthesis by glycolysis enzymes occurs by substrate-level phosphorylation: phosphate is transferred to ADP from phosphorylated organic compounds (1,3 bisphosphoglycerate or 1,3 PG and phosphoenolpyruvate or PEP)

As seen in the free energy diagram above, glycolysis extracts only a small fraction of the potential energy in a molecule of glucose.
This video gives a great overview of the process and products of glycolysis:
Pyruvate is oxidized to acetyl-coA and CO2
In respiring cells (a terminal electron acceptor is present and the electron transport chain is functioning), the two molecules of pyruvate are each:
- decarboxylated (one of the 3 carbons comes off as CO2)
- oxidized (electrons transfer to NAD+ to make NADH)
- covalently bonded to Coenzyme A (CoA) to make acetyl-CoA
All 3 steps are carried out by a single enzyme complex known as pyruvate oxidase.
- Inputs (per molecule of glucose): 2 pyruvates, 2 CoA, 2 NAD+
- Outputs (per molecule of glucose): 2 acetyl-CoA, 2 CO2, 2 NADH
Pyruvate oxidation occurs in the cytoplasm of prokaryotic cells. In eukaryotic cells, pyruvate is imported into the mitochondrial matrix for pyruvate oxidation.
This video explains what happens to pyruvate:
Acetyl-coA enters the citric acid cycle
Acetyl-CoA combines with a 4-carbon compound, oxaloacetate, to form a 6-carbon compound, citrate (citric acid) + free CoA. The citric acid cycle then releases two carbons from citric acid as carbon dioxide through a series of redox reactions that also reduce electron carriers to produce 3 NADH and 1 FADH2, 1 ATP through substrate-level phosphorylation, and oxaloacetate, ready to accept another acetyl-coA.
- Inputs (per molecule of glucose): 2 acetyl-CoA, 6 NAD+, 2 FAD, 2 ADP
- Outputs (per molecule of glucose): 2 CoA, 4 CO2, 6 NADH, 2 FADH2, 2 ATP

The citric acid cycle occurs in the cytoplasm of prokaryotic cells and in the mitochondrial matrix of eukaryotic cells.
This video gives a great overview of the process and products of the citric acid cycle:
Net ATP production per molecule of glucose oxidized
The maximum ATP production occurs during aerobic respiration. The reduced electron carriers (NADH and FADH2) dump electrons onto the electron transport chain to power chemiosmotic ATP synthesis.
In prokaryotes, the total ATP production from both oxidative phosphorylation and substrate-level phosphorylation can be as high as 38 ATP per molecule of glucose.
In eukaryotes, the ATP production is slightly lower because of the energy cost of transporting pyruvate into mitochondria: a common figure is 36 ATP per molecule of glucose.
This video gives a great overview of the process and products of the electron transport chain (this video will serve as a good refresher of concepts discussed in the last reading):
Recommended exercise
Draw your own diagram of the metabolic pathways from glucose to carbon dioxide, one for prokaryotic cells and another for eukaryotic cells, showing the sequence of pathways and where they occur in the cell. Include the electron transport chain and the ATP synthase. You can compare your diagram with a summary diagram on the next web page on fermentation.
And here’s a reminder of the BIG picture:
Protein catabolism
Where do proteins enter these pathways? Proteins are first hydrolyzed to individual amino acids. The amino group nitrogens are stripped off and sent to the urea cycle. The remaining carbon skeletons are converted to pyruvate, acetyl-CoA, or various intermediates of the citric acid cycle, depending on the amino acid. You do not need to learn any additional pathways; just realize that protein catabolism uses pyruvate oxidation and the citric acid cycle, depending on the amino acid. Various amino acids are listed in the diagram below.
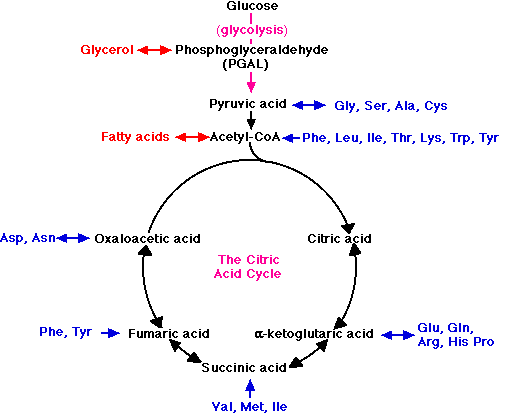
Fat metabolism
Fats are hydrolyzed to glycerol and fatty acids. Glycerol is converted to a glycolysis intermediate upstream of pyruvate, and the fatty acids are oxidized to acetyl-CoA, 2 carbons at a time. The metabolism of fatty acids, called beta-oxidation, occurs in the mitochondrial matrix and generates both reduced electron carriers and acetyl-CoA, which is further oxidized in the citric acid cycle.
This (cropped) video discusses fat and protein metabolism:
via ytCropper
For reference, you can watch the full-length video here if you’d like; start at 15:36 for discussion of fat and protein metabolism, or watch the whole thing for review on the entire reading. The last 10 minutes are a preview of the next reading, fermentation.)
Powerpoint file used in the video: oxidative_pathways_to_NADH
Sustainable Development Goals

UN Sustainable Development Goal (SDG) 2: Zero Hunger – The metabolism of glucose to carbon dioxide during cellular respiration is the primary means by which cells extract energy to support life. This process involves breaking down glucose through glycolysis, pyruvate oxidation, and the citric acid cycle, ultimately producing ATP and releasing carbon dioxide. By understanding these pathways, researchers can develop strategies to sustainably increase food production, thereby providing more food that can be used to reduce hunger worldwide.


