Learning Objectives
- Describe the main properties of the adaptive immune system.
- Identify the phylogenetic distribution of the adaptive immune system.
- Describe the role of antigen presenting cells and the distinct roles of Class I and II MHC molecules.
- Apply basic combinatorics to calculate the minimum number of possible antigen receptors for B cells or T cells.
- Compare and contrast T cell receptors and immunoglobulins as antigen receptors
- Explain immune specificity and memory in terms of the clonal selection of B cells and T cells
- Describe the overlapping but complementary roles of humoral and cellular immunity
We, and all large animals, are large bags of warm meat and broth, a rich food source not just for predators with jaws and claws, but also for the even more deadly, and ubiquitous, microbial and viral pathogens. We live because our immune cells are able to kill or neutralize these invisible predators. What is even more remarkable is that our immune cells evolve repeatedly during our lifetime. Within weeks, they evolve and adapt to fight pathogens never before seen by the individual, or encountered in previous human evolutionary history. How does this remarkable immune system work?
Innate versus adaptive immunity
All organisms, from bacteria to trees to worms to humans, have innate immune defenses. These range from physical barriers to anti-microbial chemicals. But only jawed vertebrates have evolved the complex, adaptive immune system featuring antibodies and cytotoxic “killer” cells that recognize billions of different molecules with high specificity.

Phylogenetic tree figure showing the evolution of the immune system. (Image by Olsen, V. Beth Kuser. 1997. “Evolutionary Tree.”)
Adaptive immune system has specificity and memory
The adaptive immune system takes some time: 1-2 weeks, to mount a full-fledged response to any pathogen or biological macromolecule that it sees for the first time. However, the second time it sees the same pathogen or macromolecule, it mounts an immediate, even stronger response. Both the response and the memory are highly specific to the pathogen or macromolecule.
The specificity of the adaptive immune system arises from its antigen receptors. An admittedly circular definition of an antigen is a molecule that binds to an antigen receptor: either a T-cell receptor (TCR) or an antibody molecule (immunoglobulin, Ig).
T cells and B cells generate huge variety of antigen receptors by combinatorial rearrangement of DNA segments
T cells and B cells are lymphocytes, types of white blood cells, that are able to recognize foreign (non-self) antigens. T cells have T-cell receptors on their cell surface; B cells have cell-surface antibodies. Activated B cells divide and differentiate into plasma cells that secrete large amounts of soluble antibody molecules.
Each pre-B cell or pre-T cell undergoes random combinatorial rearrangement of one copy of its immunoglobulin or TCR loci. This DNA rearrangement occurs only in differentiating B and T cell lineages. The DNA segments, called V (variable), D (diversity), and J (joining), when joined together encode the variable domain of the immunoglobulin or TCR polypeptide chain. The result is that each mature, naive (has never been activated) B or T lymphocyte expresses a unique antigen receptor on its cell surface.
Immune responses begin with antigen processing and presentation to helper T cells
A surprising finding was that antigens had to undergo processing before they could evoke an immune response. The body has professional antigen presenting cells (APCs) that have phagocytic activity and express Class II Major Histocompatibility (MHC) proteins. The most important professional APCs are macrophages, dendritic cells, and B cells. These cells phagocytose or endocytose (that is, engulf) pathogens or foreign macromolecules, digest them to peptide fragments, and then export these peptide fragments bound to MHC II proteins to the cell surface for inspection by helper T cells. Any substance that cannot be 1) processed and 2) displayed on MHC II proteins cannot provoke an adaptive immune response.
Helper T cells are activated when their TCR recognizes the antigen presented by an APC. They divide to form both activated helper T cells and long-lived memory helper T cells.
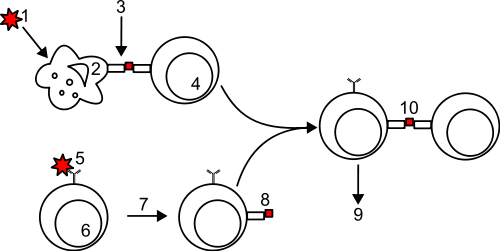
1) an antigen or pathogen is phagocytosed or endocytosed by an antigen presenting cell (APC: macrophage or dendritic cell) 2) Processing by the APC generates peptide fragments 3) Peptide fragments are bound to MHC II molecules and presented on the surface of the APC 4) A helper T cell recognizes the peptide-MHC II complex via its T-cell receptor and is activated to divide 5) A B cell binds the antigen via its cell surface antibody. 6) The antigen-antibody complex is endocytosed and processed. 7) The B cell presents processed peptide on MHC II molecules 8) An activated helper T cell recognizes the peptide:MHC II complex 10) the helper T cell activates the B cell 9) the B cell divides and produces plasma cells that secrete lots of antibody molecules
B cells recognize antigen via cell-surface immunoglobulins (antibodies)
B cells are responsible for the humoral (noncellular; mediated by soluble proteins) branch of the immune system: antibody molecules. B cells recognize and bind antigen via their cell surface Ig. They endocytose, process and present the peptide as fragments on their cell surface MHC II proteins. If these are recognized by a previously activated helper T cell, the helper T cell stimulates the B cell to divide. Dividing B cells form both plasma cells that secrete vast amounts of soluble antibody and long-lived memory B cells.
Cytotoxic T cells (CTLs) recognize and kill body cells that present foreign antigens
The cell-mediated branch of the immune system relies on cytotoxic T cells (cytotoxic T lymphocytes = CTLs). CTLs are the internal security forces. CTLs look for cells that are presenting non-self or altered peptides on Class I MHC molecules.
Most body cells express Class I MHC proteins, that present fragments of the proteins that the cells are making. Virus-infected cells will present fragments of viral proteins, and cancerous cells will present fragments of tumor proteins, which are mutated versions of normal cellular proteins.
CTLs whose T-cell receptors recognize these foreign or tumor peptide-MHC I complexes become activated and kill the virus-infected or cancerous cell. CTLs also kill cells that express a different MHC I allele than the host—this is the basis for tissue graft rejection, and why tissue and organ transplants must match at their MHC I loci.
The killing mechanism is two-fold. First, the CTL sends signals to the target cell to induce apoptosis (programmed cell death, cellular suicide). If that doesn’t work, the CTL secretes granzymes (destructive enzymes) and perforins, proteins that assemble in the target cell membrane to create holes and destroy the target cell.
Clonal selection of T-cells and B-cells lead to specific immune responses and long-term immune memory
The adaptive immune system operates on evolutionary principles of selection. The B cells and T cells have genetic diversity. Each B or T cell has a unique antigen receptor created by random combinatorial joining of their antigen receptor DNA segments. Foreign antigens selectively activate those B and T cells whose antigen receptors can bind the antigen. The B and T cells whose receptors have the highest affinity for the foreign antigen produce the most clonal progeny, all with the same antigen receptors as the parental cells. Some of these progeny become “effector” cells that activate and execute the humoral and cellular immune responses. Other progeny cells become long-lived memory B and T cells. Because the body now has an expanded population of these memory cells, the next time the same pathogen enters the body, it will be recognized much more quickly, and the immune response will be much stronger.
Powerpoint slides used in the two videos:
B1510_module5-3_ImmuneSystem_2012
Resources:
A very nice overview of the immune response, in just 6 minutes!

UN Sustainable Development Goal (SDG) 8: Decent Work and Economic Growth – Full and productive work relies on a healthy populace. By understanding how immunity works, a sustainable workforce can be protected. Having insight into the complex interplay between different components of the immune system and how they work together to protect the body helps to characterize the threats to the economy that pathogens and various diseases may pose. By considering human health interventions, such as vaccinations, we can buffer the economy against future threats caused by disease.




This may be slightly off-topic, but as T and B cells generate antigen receptors that recognize viral / bacterial proteins, is there a possibility that they have the genetic code to actually make antigens, and thus viral proteins?
This is unrelated to the immune system, but the human genome contains a number of endogenous retroviral genomes. Retroviruses infected the germline cell of ancestral humans and now all humans contain these retroviral genomes integrated into our chromosomes. Some of these still have intact genes and could make viral proteins/antigens.
Really? Do you have any sources or further reading you could link me? Very interesting
Here’s a minireview: http://genomebiology.com/content/2/6/REVIEWS1017 and here’s a recent article with evidence for activity of HERVs (human endogenous retroviruses) in human populations: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3625200/